Selecting the right pharmaceutical separation technologies is a critical decision that directly impacts product quality, regulatory compliance, and operational efficiency in pharmaceutical manufacturing. The choice involves evaluating multiple factors including the specific compounds being processed, required purity levels, production volumes, and regulatory requirements that govern pharmaceutical manufacturing processes.

The pharmaceutical industry relies on precise separation methods to ensure active pharmaceutical ingredients meet stringent quality standards while maintaining cost-effectiveness and scalability. Understanding the decision framework for pharmaceutical separation technologies enables manufacturers to optimize their processes, reduce contamination risks, and achieve consistent product specifications that meet both regulatory approval and commercial viability requirements.
Understanding Pharmaceutical Separation Technology Categories
Mechanical Separation Technologies
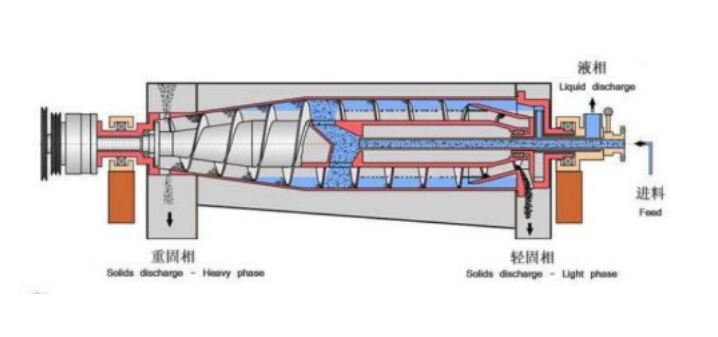
Mechanical pharmaceutical separation technologies utilize physical forces to separate particles based on size, density, or shape differences. These methods include centrifugation, filtration, and screening processes that operate without chemical reactions or phase changes. Centrifugal separators, particularly decanter centrifuges, excel at separating solid particles from liquid phases in pharmaceutical manufacturing.
The selection of mechanical pharmaceutical separation technologies depends on particle size distribution, density differences between phases, and required separation efficiency. These systems offer advantages in terms of continuous operation, scalability, and minimal chemical contamination, making them suitable for processing pharmaceutical intermediates and final products where maintaining chemical integrity is paramount.
Mechanical separation methods also provide excellent control over operating parameters such as G-force, residence time, and throughput rates. This control enables pharmaceutical manufacturers to optimize separation performance while maintaining consistent product quality and meeting regulatory requirements for pharmaceutical processing equipment.
Chemical and Chromatographic Separation Methods
Chemical separation approaches in pharmaceutical separation technologies involve selective dissolution, precipitation, or extraction processes that leverage chemical property differences between target compounds and impurities. These methods often provide higher selectivity than mechanical approaches but require careful consideration of chemical compatibility and downstream processing requirements.
Chromatographic pharmaceutical separation technologies offer exceptional precision for purifying active pharmaceutical ingredients and removing closely related impurities. These systems utilize stationary and mobile phases to achieve selective separation based on molecular interactions, though they typically operate in batch mode and require specialized expertise for optimization and maintenance.
The choice between chemical and chromatographic methods depends on the required purity levels, scale of operation, and economic considerations. While these pharmaceutical separation technologies can achieve very high selectivity, they often involve higher operating costs and more complex process control compared to mechanical separation approaches.
Evaluating Process Requirements and Specifications
Product Quality and Purity Standards
Pharmaceutical separation technologies must be selected based on the specific purity requirements established by pharmacopeial standards and regulatory authorities. The target purity level directly influences the complexity and cost of the separation system, with higher purity requirements typically necessitating multiple separation stages or more sophisticated technologies.
Understanding the nature and concentration of impurities present in the feed material is essential for choosing appropriate pharmaceutical separation technologies. Some impurities may require specific separation mechanisms, while others can be effectively removed using general-purpose separation equipment such as high-efficiency centrifuges or advanced filtration systems.
The analytical methods used to verify separation performance must also be considered when selecting pharmaceutical separation technologies. The chosen separation system should produce products that can be readily analyzed using standard pharmaceutical testing methods while meeting established specifications for identity, purity, and potency.
Production Scale and Throughput Requirements
Production volume requirements significantly influence the selection of pharmaceutical separation technologies, as different technologies exhibit varying scalability characteristics and throughput capabilities. Small-scale laboratory separations may utilize different approaches compared to large-scale commercial production, requiring careful consideration of scale-up feasibility.
Continuous versus batch operation preferences also impact the choice of pharmaceutical separation technologies. Continuous separation systems, such as pharmaceutical separation technologies utilizing decanter centrifuges, offer advantages for high-volume production while batch systems may provide better control for specialty or high-value products.
The flexibility to handle varying feed compositions and production schedules should be evaluated when selecting pharmaceutical separation technologies. Systems that can accommodate changes in feed characteristics or production demands provide operational advantages and help optimize overall manufacturing efficiency.
Regulatory and Compliance Considerations
Good Manufacturing Practice Requirements
All pharmaceutical separation technologies must comply with Good Manufacturing Practice guidelines that govern equipment design, operation, and maintenance in pharmaceutical manufacturing facilities. These requirements include specifications for materials of construction, surface finishes, cleaning procedures, and documentation systems that ensure product quality and patient safety.
The validation requirements for pharmaceutical separation technologies involve comprehensive testing and documentation to demonstrate that the equipment consistently produces products meeting predetermined specifications. This process includes installation qualification, operational qualification, and performance qualification phases that must be completed before commercial operation.
Change control procedures must be established for pharmaceutical separation technologies to manage modifications to operating parameters, maintenance procedures, or equipment configurations. These procedures ensure that any changes are properly evaluated, approved, and documented to maintain regulatory compliance and product quality.
Documentation and Traceability Standards
Pharmaceutical separation technologies require comprehensive documentation systems that track equipment performance, maintenance activities, and process parameters throughout the product lifecycle. These records must be maintained according to regulatory requirements and made available for inspection by regulatory authorities.
Traceability requirements for pharmaceutical separation technologies include maintaining detailed records of feed materials, processing conditions, and product outputs for each production batch. This information enables rapid investigation of quality issues and supports product recall procedures if necessary.
Electronic record systems used with pharmaceutical separation technologies must comply with regulatory requirements for data integrity, including provisions for secure storage, controlled access, and audit trails that track all data modifications or deletions over time.
Economic and Operational Factors
Capital Investment and Operating Costs
The total cost of ownership for pharmaceutical separation technologies includes initial equipment purchase, installation costs, ongoing maintenance expenses, and utility consumption throughout the equipment lifecycle. These costs must be evaluated against the expected production volumes and product margins to determine economic viability.
Operating costs for pharmaceutical separation technologies encompass consumables, energy consumption, labor requirements, and waste disposal expenses. Energy-efficient separation systems can provide significant cost savings over the equipment lifetime, particularly for high-throughput operations where utility costs represent a substantial portion of operating expenses.
Maintenance costs for pharmaceutical separation technologies vary significantly based on equipment complexity, operating environment, and maintenance requirements. Simple mechanical separation systems typically offer lower maintenance costs compared to complex chromatographic or chemical separation systems that require specialized technical support and replacement components.
Technology Lifecycle and Obsolescence Risk
The expected service life of pharmaceutical separation technologies should be evaluated against the anticipated product lifecycle and regulatory environment. Technologies with long service lives and established supplier support provide greater long-term value and reduced risk of obsolescence during critical production periods.
Supplier stability and technical support availability are crucial factors when selecting pharmaceutical separation technologies, as equipment manufacturers must provide ongoing support for spare parts, technical assistance, and regulatory compliance updates throughout the equipment lifecycle.
Technology upgrade paths should be considered when implementing pharmaceutical separation technologies to ensure that equipment can be modified or enhanced as production requirements evolve or new regulatory requirements are implemented without requiring complete replacement.
Implementation and Optimization Strategies
Integration with Existing Manufacturing Systems
Successful implementation of pharmaceutical separation technologies requires careful integration with existing manufacturing infrastructure, including upstream and downstream processes, utility systems, and control systems. This integration ensures smooth material flow and optimal overall process efficiency while maintaining product quality standards.
Control system compatibility is essential when integrating pharmaceutical separation technologies with existing manufacturing equipment. Modern separation systems should provide standardized communication protocols and data interfaces that enable seamless integration with plant-wide control and monitoring systems.
Operator training requirements for pharmaceutical separation technologies must be evaluated to ensure adequate expertise is available for safe and effective equipment operation. Training programs should cover normal operation, troubleshooting, maintenance procedures, and emergency response protocols specific to the selected separation technology.
Performance Monitoring and Optimization
Continuous monitoring systems for pharmaceutical separation technologies enable real-time assessment of separation performance and early detection of potential issues that could impact product quality. These systems should track key performance indicators such as separation efficiency, throughput rates, and product quality parameters.
Process optimization for pharmaceutical separation technologies involves systematic evaluation of operating parameters to maximize separation performance while minimizing operating costs and maintaining regulatory compliance. This optimization should be based on statistically designed experiments and validated analytical methods.
Regular performance reviews for pharmaceutical separation technologies help identify opportunities for improvement and ensure continued compliance with evolving regulatory requirements. These reviews should include analysis of process data, maintenance records, and product quality trends to support continuous improvement initiatives.
FAQ
What are the most critical factors when choosing pharmaceutical separation technologies?
The most critical factors include regulatory compliance requirements, product purity specifications, production scale, and total cost of ownership. The selected technology must meet Good Manufacturing Practice standards while achieving the required separation efficiency and maintaining economic viability for the intended production volumes.
How do regulatory requirements influence the selection of pharmaceutical separation technologies?
Regulatory requirements mandate that pharmaceutical separation technologies comply with Good Manufacturing Practice guidelines, including equipment validation, documentation systems, and change control procedures. These requirements often favor established technologies with proven regulatory acceptance and comprehensive vendor support for validation activities.
What is the typical validation timeline for implementing new pharmaceutical separation technologies?
Validation timelines for pharmaceutical separation technologies typically range from 6 to 18 months, depending on equipment complexity and regulatory requirements. This timeline includes installation qualification, operational qualification, performance qualification, and process validation phases that must be completed before commercial production.
How do you evaluate the long-term viability of pharmaceutical separation technologies?
Long-term viability evaluation considers technology maturity, supplier stability, maintenance requirements, and regulatory acceptance. Technologies with established track records, reliable supplier support, and compatibility with emerging regulatory trends provide better long-term value and reduced obsolescence risk for pharmaceutical manufacturing operations.
Table of Contents
- Understanding Pharmaceutical Separation Technology Categories
- Evaluating Process Requirements and Specifications
- Regulatory and Compliance Considerations
- Economic and Operational Factors
- Implementation and Optimization Strategies
-
FAQ
- What are the most critical factors when choosing pharmaceutical separation technologies?
- How do regulatory requirements influence the selection of pharmaceutical separation technologies?
- What is the typical validation timeline for implementing new pharmaceutical separation technologies?
- How do you evaluate the long-term viability of pharmaceutical separation technologies?